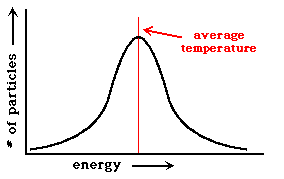
These collisions had three major results. The first was that the universe reached a condition called thermal equilibrium. To give you an idea about what this is we'll look at a glass of water at 40 degrees. The temperature of an object is a reflection of the amount of energy present in that sample of matter. However, not every molecule present has the energy that corresponds to 40 degrees. The total energy is actually spread out over a range of energies, so that there are some that have more energy than the corresponding temperature, and others that have less. This is what it looks like on a graph:

These molecules are constantly colliding with the surrounding molecules and results in exchanges of energy. This causes the number of molecules at certain energies to change. If something is in thermal equilibrium, every molecule that changes its energy level will result in another molecule changing its energy level to replace it (not necessarily in the same collision). In a way, this means that the energy present in a system is spread out among particles in such a way that the population of molecules at different energies doesn't change, even though the molecules themselves are constantly changing energy levels. In the early universe, as a result of the rapid collisions between particles, there was a state of thermal equilibrium. The reason this is so important is that things in thermal equilibrium can be quantified. This means that the system can be described by mathematical formulas, and that predictions can be made as to how the system would would change with time. Therefore, we can follow the evolution of the early universe through these formulas, even though we weren't there.
The other two consequences of these collisions involve interactions between particles as they collided.
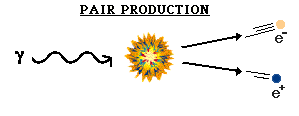
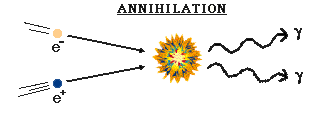
The first interaction to be considered was the constant annihilation and re-creation of electrons and positrons. One of the most famous scientific discoveries of this century is the equivalence of matter and energy. The basic concept is that under the proper conditions, energy can be turned into matter, or vice versa. This is not something common to our experience because of the conditions in which we now live (it's too cold and there's not enough pressure). But in the early universe, with its high temperature and density, this was common. Photons were converted into electrons and positrons. (Known as PAIR PRODUCTION) They could not be converted into heavier particles (protons and neutrons) because they didn't have enough energy. These electrons and positrons would eventually collide with their respective anti-particle, and then be changed back into radiation. (Referred to as ANNIHILATION)
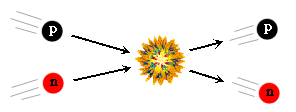
The second interaction was the conversion between protons and neutrons. These heavier atomic particles were already present In the Beginning. They were continually changing back and forth by means of the following two reactions:
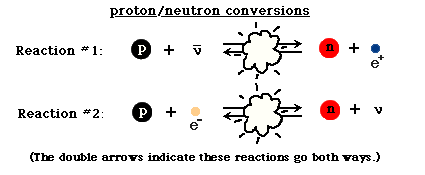
In the beginning, because of the high energy density, the collisions between particles happened so rapidly that the proton- and neutron-creating reactions balanced each other out, and the relative number of protons and neutrons, though small, were equal. But, the equality between protons and neutrons was broken almost immediately. A neutron is slightly heavier than a proton. Therefore, it requires a little more energy to change a proton into a neutron than vice versa. Initially this didn't matter because there was plenty of energy to go around. But, because the energy density was decreasing as the universe expanded, there was less energy available for each collision. This started to tip the balance in favor of the proton-forming reactions. This lead to an increase in the number of protons compared to neutrons, and as the temperature dropped more, this effect became more exaggerated. (The final numbers will be mentioned later on.)
According to the formulas, at 13.82 seconds after the Beginning, the temperature had dropped to 3,000,000,000 K. At this point there was a drastic reduction in the population of electrons and positrons. The reason for this was once again the expansion of the universe. As electrons and positrons were annihilated, the radiation that formed was stretched (specifically its wavelength) by the growing universe. This reduced the energy carried by the photons below the level which would allow them to be converted back into electrons and positrons.
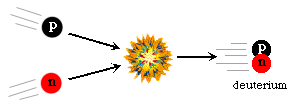
Up to this time (just over three minutes past the Beginning) there had been no nucleosynthesis. This was a result of the high energy density. In order to form atomic nuclei, the nucleons (the scientific word for protons and neutrons) must be able to collide and stick together. In the early universe the key reaction was the collision of a proton and a neutron to form a deuterium nucleus (an isotope of hydrogen). Collisions between protons and neutrons had been happening continuously since the Beginning, but their energies were too high to allow them to stick together to form deuterium nuclei.
This prevented further nuclear reactions leading to heavier nuclei. This type of situation where an intermediate product is the weak link in the overall synthesis is sometimes called a "bottleneck." This concept also applies in nucleosynthesis of heavier elements. Once the bottleneck is overcome, the remaining reactions are able to be completed. In the early universe, once the deuterium bottleneck was cleared, the newly formed deuterium could undergo further nuclear reactions to form Helium.
This could happen by means of two different reaction pathways described below.
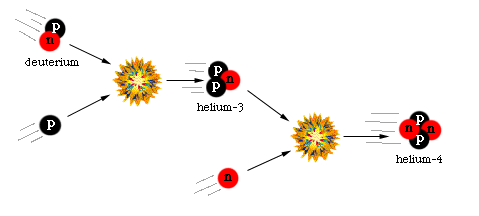
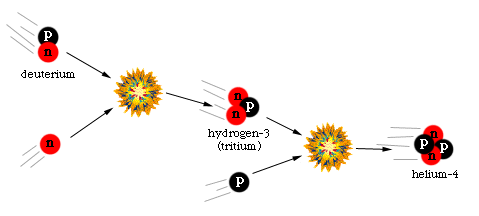
He nuclei were the heaviest to form. This was the result of the energy density being too low to allow heavier nuclei to collide with enough energy to stick. At the time that nucleosynthesis began, the relative abundance of protons to neutrons was 13% neutrons and 87% protons. When nucleosynthesis began, all the neutrons present were incorporated into He nuclei. When all the neutrons were used up, the remaining protons remained as hydrogen nuclei. So, when this first wave of nucleosynthesis was completed, the universe consisted of roughly 25% He and 75% H (by weight).
Below is a graphical summation of nucleosynthesis in the early universe. The graph shows the relative abundances of different nuclei (vertical axis) during the first three hours of creation. The horizontal axis has been labeled using both time (top) and the equivalent temperature (bottom). For those not used to using a logarithmic scale, a dashed line has been added at the 1% abundance level. Anything below this line would be less than 1% of the total mass present.
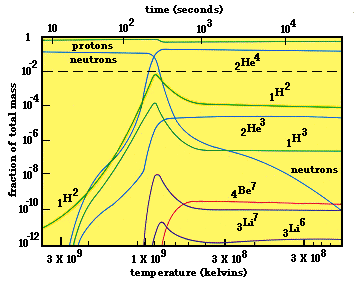
As can be seen from the curves, at the higher temperatures only neutrons and protons exist, with there being more protons than neutrons. But, as the temperature decreases, there is an increase in the amount of deuterium and helium nuclei. Just below 1 billion degrees there is a significant increase in deuterium and helium, and a decrease in the abundance of protons and neutrons. This is the deuterium bottleneck mentioned previously. This uses up the all the free neutrons and some protons, and causes the neutron line to drop off, and the proton line to dip (relatively few protons are used up). The deuterium abundance only increases to a point because it is an intermediate to the formation of helium. So as it is created, it is quickly consumed to complete the process of helium nucleosynthesis. Once all the neutrons have been used up, its presence drops off.
The final step in the formation of elements was capture of the proper number of free electrons to form neutral atoms.
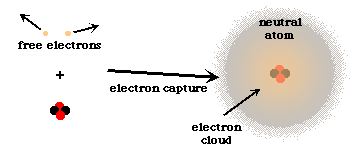
But, the remaining electrons still had plenty of energy, so it took about 700,000 years of cooling until this was able to occur. The capture of electrons to form atoms resulted in an important change in the universe. At that moment, without free electrons to interact with the photons present, the universe became transparent to radiation. This means that the photons were freely able to expand with the universe. These photons had high energies, which means that they had short wavelengths. But the expansion of the universe caused the wavelengths to get stretched out as the universe grew. These stretched out photon wavelengths are what we now refer to as the Cosmic Microwave Background (CMB). They are a leftover from the Big Bang. We have been able to measure the intensity of this background radiation, and it has closely matched that which is predicted from theoretical calculations. This has been a strong evidence in support of the "Big Bang" theory of the creation of the universe.